Introduction
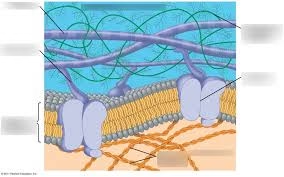
- The extracellular matrix (ECM) is the non-living part of a tissue that surrounds and supports the living cells. It is a complex network of proteins, carbohydrates, and other molecules secreted by the cells themselves.
- The ECM acts as a scaffold that helps tissues maintain their structure, provides strength, and allows cells to attach, move, and communicate with each other.
- It is found in all tissues, especially in connective tissues like cartilage, bone, and tendons, where it provides mechanical support and flexibility.
- Major components of the ECM include collagen, elastin, glycoproteins (like fibronectin and laminin), and proteoglycans.
- Overall, the extracellular matrix plays an essential role in cell growth, repair, differentiation, and tissue organization.
There are 3 types of Muscle Tissue:
Striated Muscles –
- i) Skeletal muscles- attached to skeletal system (bones).
- ii) Cardiac muscles- found in the heart
- Smooth Muscle – in walls of internal organs (e.g. stomach, small intestine) and in blood vessels.
- Most skeletal muscle, as the name implies, is attached to bone, and its contraction is responsible for supporting and moving the skeleton. The contraction of skeletal muscle is initiated by impulses in the neurons to the muscle and is usually under voluntary control.
- Contraction of the smooth muscle surrounding hollow organs may propel the luminal contents through the organ or it may regulate internal flow by changing the tube diameter. Smooth muscle contraction is controlled by the autonomic nervous system, hormones, autocrine/paracrine agents and other local chemical signals.
- Cardiac muscle is the muscle of the heart. Its contraction propels blood through the circulatory system. Like smooth muscle, it is regulated by the autonomic nervous system, hormones, and autocrine/paracrine agents and it can undergo spontaneous contractions. Although there are significant differences in these three types of muscle, the force-generating mechanism is similar in all of them.
Structure of muscle
1) Skeletal muscles-
- The cells of skeletal muscles are cylindrical in shape.
- Each cell is commonly called muscle fiber.
- Each muscle fiber is multinucleated (syncytical) cell. Each cell is surrounded by an electrically excitable plasma membrane, the sarcolemma.
- Each of the muscle fibers are made of many myofibrils, held together by connective tissue.
- Myofibrils are composed of bundles of myofilaments.
- Myofilaments consist of thick filament and thin filament.
– The thick filament consists of protein myosin.
– The thin filament consists of protein actin, tropomyosin and troponin.
- Myofilaments are the actual contractile elements of striated muscle.
- When myofibril is examined by electron microscope alternating dark A (anisotropic) bands and light I (isotropic) bands can be observed.
- The central region of the A-band termed the H band is less dense than the rest of the band.
- The I (light) band is bisected by a very dense, narrow Z-line or Z-disks. The Z-line is made of the protein α-actinin and desmin.
- The portion of the myofibril that lies between two successive Z-line is called sarcomere, i.e. sarcomere, is a Z-line to Z-line repeat which repeats every 2.3 mm (2300 Å) along the fibril axis.
- Sarcomere represents the smallest functional unit of myofibril. It is the basic contractile unit of striated muscle.
- The thick and thin filaments of sarcomere do not change length or width during muscle contraction.
- During muscle contraction, the thick and thin filaments overlap each other. Consequently, the H bands and I bands shorten.
- The length of the sarcomere which is 2300 nm in an extended form of myofibril is reduced to 1500 nm in a contracted form.
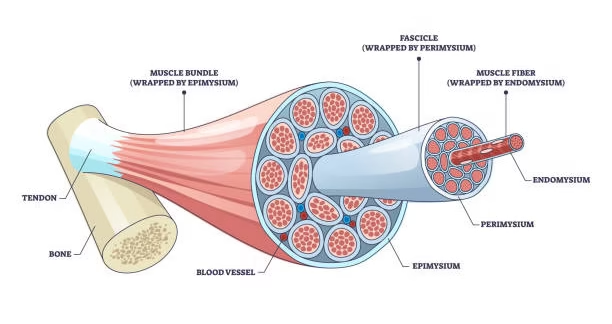
Fig: A: Skeletal muscle;
B: Muscle fiber composed of myofibrils;
C: Myofibril composed of myofilaments,
D: Arrangement of myofilaments in striated muscle
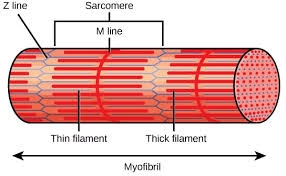 Fig: Arrangement of muscle filaments
Fig: Arrangement of muscle filaments
Where A: Relaxed or extended position; B: Contracted position
Protein composition of muscle fiber
Muscle contains following types of proteins:
- Contractile proteins
- Myosin
- Actin.
- Regulatory proteins
- Tropomyosin
- Troponin.
- Minor or accessory proteins.
- Contractile proteins
Myosin
i) Each myosin molecule is composed of six polypeptide chains, two identical heavy chains and four light chains. Light chains are of two types.
ii) Two identical heavy chains are wound around each other to form a double helix, which is called the tail of the myosin molecule.
iii) The amino terminal end of each of these chains is folded into a globular structure called the myosin head. Thus, there are two free heads lying side-by side at one end of the double helix myosin molecule.
iv)One each type of light chain is associated with each of the myosin heavy chain heads. These light chains help to control the function of the head during muscle contraction.
v) Globular head of myosin has two specific binding sites, one for ATP and one for actin. lt also exhibits ATPase activity which can hydrolyze ATP to ADP and Pi.
vi) Tails of the myosin molecules bundled together to form the thick filament, while many heads of the myosin molecule hang outward to the sides of the thick filament except H-region, where there are no myosin heads.
Functions of myosin
Myosin has three important functions as follows:
- Constituent of thick filament of muscle fiber.
- The amino terminal globular ends of myosin exhibit adenosine triphosphatase (ATPase) activity. It hydrolyzes ATP to ADP + Pi and provides free energy for muscle contraction.
- Myosin interacts with actin and generates the force (power stroke) that moves the thick and thin filament past each other.
Actin
i) Actin is the major constituent of thin filament of muscle fiber.
ii) Actin is a polymer of globular-shaped subunit called G-actin.
iii) In presence of Mg2+ ions, G-actin polymerizes spontaneously into a fibrous or filamentous form called F-actin.
iv) Each G-actin molecule of the thin filament has a binding site for myosin.
v) In association with myosin, actin plays a major role in muscle contraction.
Regulatory proteins
Tropomyosin
i) Tropomyosin is a constituent of thin filaments of the muscle.
ii) Tropomyosin is made up of two polypeptide chains, which is wrapped spirally arround the sides of the F-actin helix.
iii) In the resting state, the tropomyosin molecules lie on top of the active sites of the actin strands, so that attraction cannot occur between actin and myosin filaments to cause contraction.
iv) Tropomyosin is involved in the contraction process by regulating the attachment of actin and myosin.
Troponin
i) Troponin is also a constituent of thin filaments of the muscle.
ii) Troponin is a complex of three polypeptide chain, TnC, TnI and TnT, which is attached to tropomyosin and each of which plays a specific role in controlling muscle contraction as follows:
TnC (Troponin C): It binds calcium ions, the essential step in the initiation of muscle contraction.
TnI (Troponin I): It binds to actin and inhibits actin-myosin attachment.
TnT (Troponin T): It binds to tropomyosin, anchoring the troponin complex.
Minor or Accessory proteins of Myofibrils
They are involved in stabilizing the structure and function of muscles.
Mechanism & biochemical events of muscle contraction
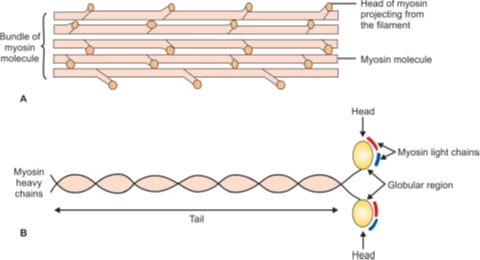 Image A: The thick filament consists of bundles of myosin molecules;
Image A: The thick filament consists of bundles of myosin molecules;
Image B: Schematic structure of myosin molecule
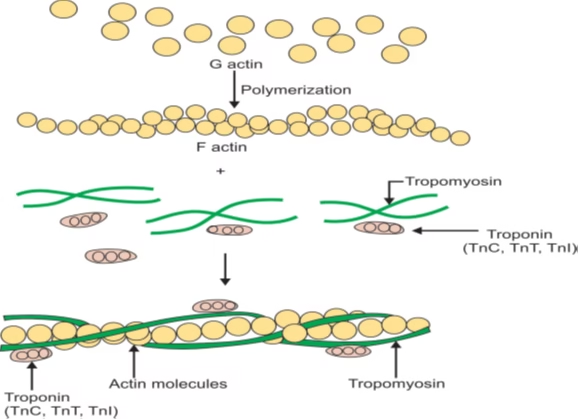 Image 3: Actin, Troponin & Tropomyosin
Image 3: Actin, Troponin & Tropomyosin
Biochemical basis/Mechanism of Muscle contraction
“Sliding filament model”, is the most accepted model of muscle contraction proposed in 1954. According to this mechanism, the thin filaments slide past the thick filaments during contraction so that the total length of the fiber is shortened. Although the length of the sarcomere decreased during muscular contraction, the lengths of the individual thick and thin filaments did not change but the H-zones and I-bands shortened.
Biochemical Events Occurring During Muscle Contraction
Each contraction cycle consists of five stages as follows:
- Binding of ATP to head of myosin of thick filaments and detachment of myosin from actin (thin filament).
- Conformational changes in the myosin head as a result of hydrolysis of ATP to ADP+Pi.
- Binding of myosin-ADP-Pi complex to actin.
- Force generation in which myosin head releases inorganic phosphate and power stroke occurs.
- Reattachment of myosin head to the new actin molecule of the thin filament and formation of rigor complex.
Stage 1.
i) In the first stage, ATP binds to the myosin head of the thick filament which is tightly bound to the actin molecule of the thin filament
ii) Binding of ATP dissociates actomyosin into actin and myosin
iii) Binding of ATP induces conformational changes in the myosin head. This change reduces the affinity of the myosin head for the actin molecule of the thin filament causing the myosin head to detach from the thin filament.
Stage 2.
i) Adenosine triphosphatase (ATPase) of the myosin hydrolyzes ATP to ADP and Pi and remain bound to myosin head.
ii) The energy released by the splitting of ATP is stored in the myosin molecule.
iii) Myosin in the form of myosin ADP-Pi complex is now in a high energy state. This is the predominant state at rest, i.e. relaxed state.
Stage 3.
i) Upon muscle stimulation via calcium, the inhibition of actin-myosin attachment imposed by the regulatory proteins (tropomyosin and troponin) is removed and consequently the myosin with bound ADP and Pi attaches to actin.
ii) It is believed that the angle of myosin head attachment is 90°.
Stage 4.
i) The actin-myosin reassociation stimulates the release of inorganic phosphate (Pi) from myosin head; which results in increasing the strength of the myosin-actin attachment.
ii) This tight binding of myosin to actin results in a conformational change in myosin head, tilting the angle of head from 90° to 45°.
iii) This change in myosin head, causes, the generation of force called the ‘Power Stroke’ of muscle contraction which pulls the thin filament a distance about 70°A (10 nm) towards the center the sarcomere.
Stage 5.
The myosin head is again tightly bound with release of ADP to a new actin molecule of the thin filament which is known as rigor configuration.
-
- The actin-myosin complex remains intact until another molecule of ATP binds to the head of myosin molecule.
- With a new ATP, a new cycle may begin and the cycling may continue as long as regulatory calcium and ATP are present.
Image: Biochemical events occurring during muscle contraction.
The immediate source of energy in muscle for contraction is ATP. ATP can be generated from the following ways:
– By substrate level phosphorylation of Glycolysis using glucose or muscle glycogen
– By oxidative phosphorylation
– From creatine phosphate.
Muscle disorders
Muscular Dystrophy
- Muscular dystrophies are a group of genetic conditions characterized by progressive muscle weakness and wasting without involvement of nervous system.
- This is an X-linked disorder and caused by mutations in the gene coding for the protein dystrophin.
- The different types of muscular dystrophy affect different sets of muscles and result in different degrees of muscle weakness.
- The commonest and best characterized types are:
– Duchenne muscular dystrophy (DMD)
– Becker muscular dystrophy (BMD)
- Duchenne and Becker types of muscular dystrophy primarily affect the skeletal muscles, which are used for movement, and the muscles of the heart.
Structure & Associated Disorders of Connective tissue
Connective Tissue (Extracellular Matrix)
- It refers to complex material surrounding the mammalian cells in tissues.
- It is predominantly made up of fibrous proteins like collagen, elastin, keratin, fibrillin, fibronectin, laminin and glycoconjugates like proteoglycans.
Collagen
- Collagen is the most abundant protein in mammals, comprising approximately one third of the total body protein.
- It is the chief constituent of connective tissue.
- Its distribution varies in different tissues; collagen forms 90% of the organic matrix of bones, 85% of tendons, 70% of skin and 4% of liver.
- Collagen occurs in about 19 distinct forms coded by 30 different genes.
- Type I Collagen: most abundant in all regions of the body
- Type II Collagen: mainly in cartilage and vitreous humour.
- Type III Collagen: in skin, lungs and vascular tissue.
- Type IV Collagen: in basement membranes
Functions of Collagen:
- Collagen gives strength, support and shape to the tissues.
- Collagen contributes to proper alignment of cells, which in turn helps in cell proliferation and their differentiation to different tissues and organs.
- Collagen (that is exposed in blood vessels) contributes to thrombus formation.
Structure of collagen:
- Collagen is organized in a triple helical form.
- Every third position of triple helix is occupied by Glycine.
- Collagen is composed of repeating structures of (Gly-X-Y)n where X, Y represent proline, hydroxyproline and sometimes hydroxylysine.
- Hydroxylation of proline and lysine occurs as post-translational modification. Prolyl hydroxylase and lysyl hydroxylase both contains iron at its active site and requires ascorbic acid (vitamin C) to preserve iron in the reduced ferrous state.
- Extensive covalent cross-links between lysine and hydroxylysine residues strengthen collagen fibers. Cross-links are formed by lysyl oxidase which converts these amino acids into aldehydes. These are called as Aldol cross links.
- Lysyl oxidase is a copper containing enzyme. In copper deficiency, collagen cross-links are reduced.
- With advancing age, the extent of these cross-links increase. The process makes the skin and blood vessels more rigid and less elastic. This causes several heath complications during old age.
Collagen Diseases
Diseases associated with impaired synthesis of collagen include scurvy, lathyrism, osteogenesis imperfecta, Ehlers-Danlos syndromes and epidermolysis bullosa.
Scurvy:
Vitamin-C is essential for hydroxylation of proline and lysine to form hydroxyproline and hydroxylysine, respectively, which are integral components of the structure of collagen. In vitamin-C deficiency (scurvy), collagen synthesis is adversely affected.
Lathyrism:
It occurs due to regular intake of pulse, lathyrus sativa or sweet pea (kesari dal). It contains a toxin called beta oxalyl aminoalanine (BOAA) which inhibits the enzyme lysyl oxidase. This adversely affects the cross linking of lysine in collagen structure. As a result the bones become weak and brittle.
Osteogenesis imperfecta (brittle bone disease):
In this disease, there are mutations in the gene encoding type I collagen which affect many tissues resulting in brittle bones leading to many fractures in infancy and childhood.
Alport Syndrome:
It is genetic disorder affecting the synthesis of type-IV collagen found in the basement membranes of the renal glomeruli. The early sign is hematuria (blood in the urine) and the patient finally develops end-stage renal disease.
Ehlers-Danlos syndromes:
Due to defective type III collagen. This group of disorders is characterized by loose skin, hyper extensibility of the joints and skin.
Epidermolysis bullosa:
The condition showing blisters and cracks in the skin is caused by mutations in the gene encoding type VII collagen.
Menke’s disease:
A deficiency of copper results in defective function of lysyl oxidase, and reduced cross-linking of collagen results.
Elastin
- It confers elasticity and extensibility to arteries and tendons.
- It is found in large quantities in lungs, arterial blood vessels and elastic ligaments.
- It contains glycine, proline, alanine. Triple helix structure is absent.
- It contains desmosine and iso-desmosine cross links.
Elastin diseases:
Williams syndrome: Occurs due to deletion in elastin gene.
Scleroderma: Skin disease occurs due to accumulation of elastin.
Fibrillin, Fibronectin and Laminin:
-
- Fibrillin is located in myofibrils and is associated with elastin. Mutations in the gene for fibrillin cause Marfan’s syndrome. Its features are Arachnodactyly (long digits), ectopia lentis (dislocation of lens), hyperextension of joints, aortic aneurysm.
- Fibronectin is a glycoprotein. It plays a role in cell adhesion and migration.
- Laminin is a component of basal laminae. (Eg in renal glomerulus)
Keratin:
They are Cysteine-rich proteins mostly seen in hair and nails.
Proteoglycans
- They are conjugated proteins containing glycosaminoglycans. (GAGs)
- Ground substance of extracellular matrix is mainly composed of GAGs.
- Hyaluronic acid-
- Found in synovial fluid of joints & vitreous humor of eyes.
- Present in connective tissues and gel around ovum.
- Lubricant and shock absorbent in joints.
- Heparin-
- Anticoagulant; occurs in blood, lung, liver, kidney.
- Helps in release of lipoprotein lipase clearing lipemic plasma.
- It prevents intravascular clotting in thromboembolisms.
- Chondroitin sulfates-
- Major constituent of bone, cartilage, tendons, heart valves.
- Contributes to the compressibility and weight bearing capacity of the cartilage.
- Dermatan sulfate-
- Mostly occurs in skin & sclera.
- Responsible for maintaining shape of the eyeball.
- Keratan sulfate-
- Occurs in cornea, tendons, cartilage.
- Role in corneal transparency.
-
Pnemonics
1. Components of Extracellular Matrix
The ECM has 3 main components:
Fibers, Ground substance, and Adhesion proteinsMnemonic: “Funny Grand Aunt”
-
F – Fibers (Collagen, Elastin, Reticular)
-
G – Ground substance (Proteoglycans, Glycosaminoglycans)
-
A – Adhesion proteins (Fibronectin, Laminin, Integrins)
2. Types of Fibers in ECM
There are three main types of fibers.
Mnemonic: “CRE” – Collagen, Reticular, Elastic
-
C – Collagen fibers → Strength
-
R – Reticular fibers → Support (delicate framework)
-
E – Elastic fibers → Elasticity
3. Types of Collagen
There are many types, but focus on the first four (I–IV).
Mnemonic: “Be(So) Totally Cool, Read Books”
-
I – Bone, Skin, Tendon → most abundant
-
II – Cartilage (carTWOlage)
-
III – Reticular fibers, Blood vessels
-
IV – Basement membrane (“Floor = IV”)
4. Ground Substance Components
Ground substance = GAGs + Proteoglycans + Glycoproteins
Mnemonic: “Great People Give Support”
-
G – Glycosaminoglycans (GAGs)
-
P – Proteoglycans
-
G – Glycoproteins
-
S – Support + structure
5. Functions of Extracellular Matrix
Mnemonic: “SACRED”
-
S – Support to cells & tissues
-
A – Adhesion (helps cells attach)
-
C – Communication between cells
-
R – Regulates cell behavior (growth, differentiation)
-
E – Elasticity & resilience
-
D – Diffusion of nutrients & waste
6. Basement Membrane Components
Mnemonic: “LACE”
-
L – Laminin
-
A – Agrin
-
C – Collagen type IV
-
E – Entactin (nidogen)
7. Cells That Produce ECM
Mnemonic: “Funny Chubby Cells”
-
F – Fibroblasts (main producers)
-
C – Chondroblasts (cartilage)
-
C – Osteoblasts (bone)
-